[ad_1]
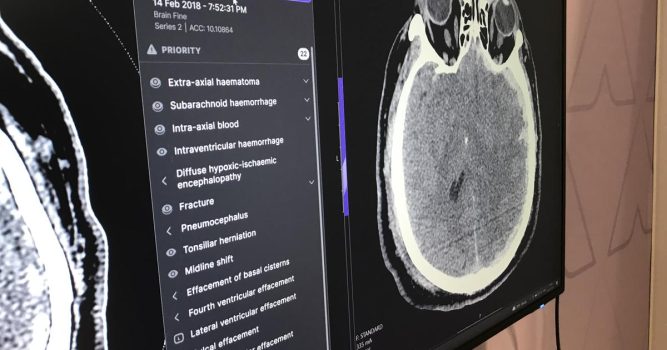
The U.S. Food and Drug Administration just granted Annalise.ai seven additional 510(k) clearances for the company’s suite of AI-assisted triage and notification solutions.
The Australia-based company made the announcement on April 12 in a release that described the timing of these AI-assisted solutions as “increasingly important” amid growing workloads and staffing shortages.
“Triage solutions drive quality improvement by enabling earlier detection and intervention for our most critically ill patients. And by prioritizing the radiology work list, these solutions also help…
[ad_2]
Go to publisher site for the complete article:
Read More